What regulates nitrate leaching into groundwater
Oaks ’n’ Folks – Volume 19, Issue 1 – February 2003
One critical issue that has long been of concern in California is water. We live in a dry state, and the demands on our supplies of surface water and groundwater are intense. We use our water supplies for agriculture, gardening, household use, and we still need to maintain adequate stream and river flow to support fisheries and natural stream ecosystems. One aspect of the larger water issue is water quality, with a growing concern about non-point-source contamination, where contaminants don’t come from a single source (e.g. outflow from a factory or waste treatment plant) but rather enter natural water supplies from diffuse sources such as agricultural fields or open land.
Nitrate (NO3–) is one such non-point-source pollutant that is common in California groundwater and surface waters. The EPA limit for drinking water is 10 parts per million of nitrate, and high nitrate concentrations in stream water can cause harmful algal blooms and stream eutrophication (buildup of nutrients). Even in areas that are not in fertilized agriculture, nitrate concentrations in California groundwater can be high enough to be a concern. The factors that lead to elevated nitrate in groundwater and stream water are not well understood. Some research has suggested that cattle are responsible. Cattle remove plants that can take up nitrate. They also urinate and defecate, producing “hot spots” of very high nitrogen availability that can overload the plant and soil microorganisms’ ability to take up that nitrogen. The excess nitrogen is converted to nitrate by other bacteria in the soil, and the nitrate can then be leached (i.e. lost from the soil) into groundwater. However, it has not really been well established that this is the mechanism producing elevated nitrate in groundwater in grazed areas in California.
With support from the Integrated Hardwood Range Management Program, my research group has been examining the factors that make California grassland ecosystems “leaky”, that is prone to leaching nitrate into groundwater. To understand this requires understanding multiple components of the nitrogen cycle: How fast is nitrogen released as ammonium from soil organic matter by microbes during decomposition? How fast is that ammonium taken up by plants? How much is converted to nitrate by nitrifying bacteria? How much of that nitrate is actually leached below the rooting zone? We need to understand the factors that regulate these processes as well. How are they influenced by things such as cattle grazing, landscape structure, and the nature of the plant community? There are two major communities that dominate most of California’s rangeland: open annual grassland and oak woodland, where there is a nearly complete canopy of oak trees. We also need to consider how the strong seasonality associated with our Mediterranean climate affects the processes and the interactions among them.
Our work is being done at the UC Santa Barbara Sedgewick Reserve, which is part of the UC Natural Reserve System. Other researchers from UC Santa Barbara and California Polytechnic at San Luis Obispo have set up various grazing manipulations at the Reserve that we used to examine plant species and grazing effects on soil nitrogen cycling processes. We are approaching this project through a variety of methods, including field measurements of nitrate in soil and groundwater, laboratory experiments to understand the controls on nitrogen cycling, and finally field experiments, including use of nitrogen-15, a non-radioactive isotope of nitrogen (the normal form is nitrogen-14), to trace the fate of nitrogen in the field. We have carried out these measurements in both open grassland and closed canopy woodland to examine how plant community affects nitrogen cycling, in grazed and ungrazed plots in both grassland and woodland sites, and in different landscape positions where soil depth and hydrology vary.
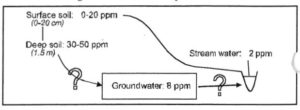
One interesting result we found is that even in ungrazed areas, during the fall and winter, soil nitrate levels can be high. Immediately around the first rains, the surface soil nitrate levels are particularly high; however by mid-winter, they decline somewhat, while NO3– in the deeper soils can increase to as much as 50 ppm (Figure 1). However, the groundwater is only around 8 ppm and stream water is around 2 ppm. These results raised the following questions: What causes the high nitrate levels in the soils in the fall and early winter? Why do the concentrations decline down the flow path from soil water to groundwater to streams?
|
|
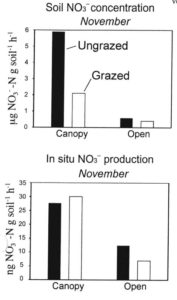
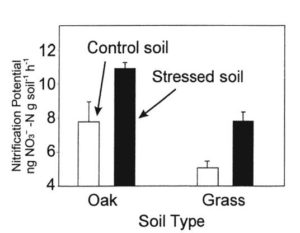
When we compared nitrogen cycling dynamics in grazed and ungrazed plots, we did not find any evidence of either higher nitrate concentrations or of higher rates of nitrate production in the grazed plots than in the ungrazed plots (Figure 2). This suggested that it was something about the seasonal transition that led to the elevated nitrate in the soils in the fall and early winter, rather than an effect of cattle grazing.Possibly, the episodic drying and rewetting that occurs in the early winter might stimulate nitrate production in these soils. It is known that such drying/rewetting cycles can be a major stress on microorganisms that can cause them to rupture, spilling their nitrogen-rich cell guts. This could stimulate other bacteria that survive the stress and provide nitrogen for nitrifying bacteria. Experiments examining the effect of multiple drying/rewetting stress cycles show that nitrifying bacteria actually do very well under these conditions (Figure 3), though they are a major stress for other bacteria. Thus, it appears that the high rates of nitrate production in the rangeland soils at Sedgwick are a natural result of the climate regime rather than due to any specific human intervention. It is possible that early season nitrate production followed by wet season leaching has caused California to always have somewhat high levels of nitrate in its groundwater and stream water.An alternative hypothesis, however, is that the leaching of nitrate into groundwater may be because the grasses that currently dominate the ecosystems are annual. Until well into the winter growing season, these grasses have very limited root systems and very limited ability to take up nitrate that may be available in the soil, allowing that nitrate to leach. It is possible that the native perennial grass vegetation that dominated these ecosystems pre-European-colonization had root systems that could become active early in the wet season and take up nitrate, preventing it from leaching into groundwater and being lost from the ecosystem. If this hypothesis is correct, then the elevated nitrate in groundwater may be a result of cattle grazing, but it would have been the heavy grazing that occurred over a century ago and facilitated the transition from native perennial grasslands to annual grasslands.
We are testing the annual-grassland leaching hypothesis by working with Dr. Reichman and Dr. Seabloom in experimental plots they have set up at Sedgwick, where they have established stands of either entirely native perennial grasses or entirely introduced annual grasses. We are applying nitrogen-15 labeled ammonium to the soils in the fall and then following the nitrogen tracer into plants and soil microorganisms, as well as determining how much leaches out of the plots. We will be able to analyze whether the fate of nitrogen really is different in the two plant community types. This work has led to the development of new theories about the regulation of ecosystem nitrogen cycling. We are also expanding our research into the role of rapid seasonal transitions in regulating nitrogen cycling and ecosystem processes in other ecosystem types as well, including chaparral and alpine tundra.We started this project by targeting a clear applied problem that has important implications for managing California rangeland. Our results suggest that the high nitrate levels in rangeland groundwater may not simply be a direct result of current cattle grazing activities. Analyzing the mechanisms responsible for nitrate leaching has required a combination of field measurement and highly sophisticated basic research, as has become common in modern applied research.
Josh Schimelprepared and edited by Adina Merenlender and Emily Heaton